Buteyko Australia, Queensland Asthma Foundation – Effect of Buteyko in the management of asthma.
Abstract
Objective: To evaluate the effect of Buteyko breathing techniques (BBT) in the management of asthma.
Design: Prospective, blinded, randomised study comparing the effect of BBT with control classes in 39 subjects with asthma. The study was conducted from January 1995 to April 1995.
Participants and setting: Subjects recruited from the community, aged 12 to 70 years, with asthma and substantial medication use.
Main outcome measures: Medication use; morning peak expiratory flow (PEF); forced expiratory volume in one second (FEV1); end-tidal (ET) CO2; resting minute volume (MV); and quality of life (QOL) score, measured at three months.
Results: No change in daily PEF or FEV1 was noted in either group. At three months, the BBT group had a median reduction in daily beta2-agonist dose of 904 µg (range, 29 µg to 3129 µg), whereas the control group had a median reduction of 57 µg (range, – 2343 µg to 1143 µg) (P = 0.002). Daily inhaled steroid dose fell 49% (range, – 100% to 150%) for the BBT group and 0 (range, – 82% to +100%) for the control group (P = 0.06). A trend towards greater improvement in QOL score was noted for BBT subjects (P = 0.09). Initial MV was high and similar in both groups; by three months, MV was lower in the BBT group than in the control group (P = 0.004). ET CO2 was low in both groups and did not change with treatment.
Conclusion: Those practising BBT reduced hyperventilation and their use of beta2-agonists. A trend toward reduced inhaled steroid use and better quality of life was observed in these patients without objective changes in measures of airway calibre.
Introduction
Proponents of Buteyko breathing techniques (BBT) suggest that hyperventilation resulting in alveolar and airway hypocapnia is a major contributor to the pathophysiology of asthma.1 Advocates claim that exercises designed to reduce minute volume lead to improvements in asthma control and allow reduction of medication.1 To our knowledge, no formal study of the techniques has been reported in accessible publications.
Aside from considerations of hyperventilation, a variety of breathing and relaxation techniques have been advocated for the complementary control of asthma. These include yoga,2, 3 diaphragmatic breathing4 and progressive muscle relaxation.5 Although varying claims of efficacy have been made, no technique has been adopted by mainstream medical practice.
We undertook a blinded, prospective, controlled, randomised study in people with asthma, in which we compared BBT with a placebo breathing technique, and measured lung function, medication use and quality of life.
Methods
Participants
Subjects aged 12 to 70 years were recruited following a publicity campaign about breathing techniques in asthma by the Asthma Foundation of Queensland. People telephoning the Foundation enquiring about BBT were referred to the investigators. Individuals were accepted if they reported a history of asthma (variable difficulty in breathing, wheeze or chest tightness with response to beta2-agonist) and were taking substantial doses of asthma medication. To match the BBT and control groups for symptomatic asthma severity, trial participants were stratified by whether they were taking more or less than 5000 µg of salbutamol or equivalent a day, and were randomly allocated to treatment groups on the basis of a sealed envelope prepared by a clerical staff member not involved in the study. Trial participants, investigators and attending staff were blinded to which group randomisation had occurred; the term Buteyko was not used at any time by either control or BBT instructors.
Subjects were excluded if there had been a change in inhaled steroid dose or use of oral steroids within the four-week run-in period, if there were other significant unstable medical conditions, or if they had undertaken BBT previously. Subjects were randomised if they were using at least 1400 µg of short acting beta2-agonist or equivalent doses of nebulised or long acting beta2-agonist in the last week of the run-in period. Nebulised beta2-agonist was considered one-fifth as effective as a dose delivered by metered dose inhaler.6 Standard doses of terbutaline and salbutamol were considered equipotent. Two doses of salmeterol (25 µg) via metered dose inhaler twice a day (100 µg/day) was considered equivalent to 200 µg of a short acting beta2-agonist every four hours (1200 µg/day).7
Intervention
Trial participants underwent training simultaneously in two separate groups. Teaching occurred over seven days; each session lasted 60-90 minutes.
BBT was taught by a representative of Buteyko Australia. Buteyko training consisted of the teaching of a series of exercises in which subjects reduced the depth and frequency of respiration. Breath holding exercises measured the impact of this training and gauged progress. Participants were encouraged to practise these exercises several times a day. The BBT instructor made follow-up phone calls to participants as she considered necessary, and people experiencing difficulty with the technique were given extra breathing classes, in accordance with standard Buteyko practices.
In the control group, subjects were given general asthma education and relaxation techniques, and were taught abdominal breathing exercises which did not involve hypoventilation. The control group instructor made one follow-up phone call to each participant.
In both groups subjects were directed to use beta2-agonists only when symptomatic. Subsequently, we telephoned the participants from both groups fortnightly and reiterated the beta2-agonist advice. Subjects were advised to reduce long acting bronchodilator dose only when short acting therapy was used once a day or less.
At clinic review, six weeks after tuition, subjects using one dose a day or less of short acting beta2-agonists were directed to reduce their inhaled steroid dose. Further review and, where possible, inhaled steroid reduction was undertaken every two weeks by telephone.
The study design is summarized in Figure 1, below.
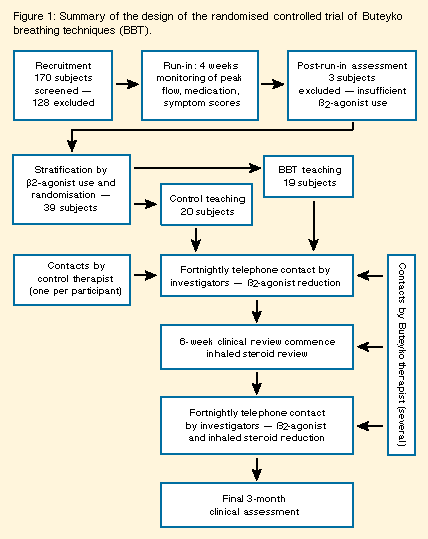
Outcome measures
Patients kept diary cards at home for scoring symptoms (0 = no symptoms, 3 = maximal symptoms) and recording daily peak expiratory flow (PEF) and medication use.
At clinic visits at the end of the run-in period and three months later, flow volume loops were recorded before and after administration of bronchodilator, and patients completed a self-administered quality of life questionnaire.8, 9 The questionnaire measured the effect of asthma on quality of life across four domains: breathing, mood, social interaction and concerns for the future.
At each clinic visit, indices of resting ventilation were measured. Subjects were told these techniques were to “assess breathing patterns” and were asked to “breathe normally”. End-tidal (ET) CO2 (as a surrogate for arterial CO2) was recorded using continuously sampled nasal expired air, aspirated by nasal cannula and analysed using infra-red absorption (POET, Sensormedics, Wawkesha, USA). Minute volume (MV) was recorded with a 5 L water-sealed spirometer with a CO2 absorber using a mouthpiece and noseclip (Gould 2400, Sensormedics, Yoba Linda, USA).
Twenty non-smoking healthy subjects age and sex matched with subjects with no history of upper or lower respiratory tract disease or other illness were recruited from hospital staff to provide comparative data for ET CO2and MV.

Statistical analysis
Data were analysed using Statview.10 Paired data were compared using paired t test for normally distributed data, and Wilcoxon signed-rank test for non-parametric data. For unpaired data, unpaired t tests and Mann-Whitney U tests were used for parametric and non-parametric data, respectively. For non-parametric data Spearman’s rank was used to investigate correlation. Data are expressed as median (range), or mean ± standard deviation.
Ethical approval
The Mater Adult Hospitals Ethics Committee approved the protocol, and informed consent was obtained from all subjects.
Results
Participants
One hundred and seventy patients were screened for the study. Most proved either not to have asthma or to have unstable asthma as described above. Thirty-nine patients were randomised, 20 to the control group and 19 to the BBT group. No significant differences existed between the groups (Box). One person from each group was lost to follow-up after the intervention.
Twenty normal subjects (10 women) were recruited; their median age was 43 years (range, 25-68).
Follow-up calls
BBT subjects each received a mean of seven (range, 0-20) follow-up telephone calls from the instructor, and extra classes were scheduled for seven BBT subjects who had difficulty with the technique.
Indices of airway function
There was no difference between the groups in measures of airway function. Morning pre-bronchodilator PEF was similar at both run-in (BBT, 385 ± 90 L/min; control, 375 ± 117 L/min; P = 0.3) and at three months (BBT, 374 ± 115 L/min; control, 383 ± 103 L/min; P = 0.6). Pre-bronchodilator percentage predicted FEV1 was also no different at run-in (BBT, 75% ± 17%; control, 73% ± 19%; P = 0.4) and at three months (BBT, 72% ± 22%; control, 72% ± 15%; P = 0.4).
Indices of resting ventilation
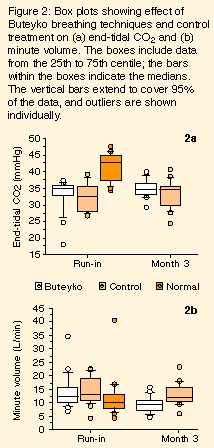
End-tidal CO2: No significant difference in mean ET CO2 existed between BBT and control groups either at run-in (BBT, 33 ± 5 mmHg; control, 32 ± 4 mmHg) or at three months (BBT, 35 ± 3 mmHg; control, 33 ± 3 mmHg). The normal subjects had significantly higher mean ET CO2 levels (41 ± 4 mmHg) than both the BBT and the control groups (Figure 2a).
Minute volume: The normal subjects recorded a mean MV of 11.9 ± 0.8 L/min. Despite a trend (Figure 2b), there was no significant difference between these subjects and the BBT and control groups either at run-in (BBT, 14.0 ± 6.5 L/min; control, 14.2 ± 4.9 L/min) or at three months (BBT, 9.6 ± 3.1 L/min; control, 13.3 ± 4.0 L/min). At three months the mean MV for the BBT group was significantly less than for the control group (P = 0.004).
The relative reduction in beta2-agonist use in the BBT group was related to the proportionate reduction in minute volume (r = 0.51; P = 0.04)
Medication use
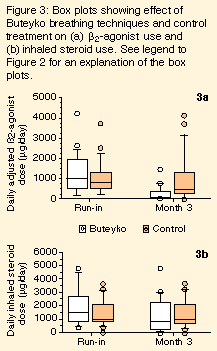
Three months after intervention, there was a significant difference in beta2-agonist use between the BBT group and the control group (P = 0.005). The BBT group had a median reduction in daily adjusted beta2-agonist dose of 904 µg (range, 29 µg to 3129 µg) and the control group had a median reduction of 57 µg (range, – 2343 µg to 1143 µg) (P = 0.002) (Figure 3a).
Despite a trend in inhaled steroid use (Figure 3b), there was no significant difference between the groups in absolute daily doses or change in dose at any time. The median reduction in daily dose at the third month, compared with run-in, was 49% (range, – 100% to + 83%) for BBT subjects and 0 (range, – 82% to + 100%) for control subjects (P = 0.06).
Serious adverse events
During the study, three subjects from each group were admitted to hospital for exacerbations of asthma. Apart from these, six BBT and seven control subjects received short courses of prednisone at some stage during the eight months after intervention.
Quality of life
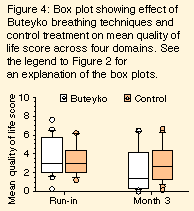
No significant difference existed between the mean quality of life score of the two groups at any stage (Figure 4). At three months, there was a trend towards greater improvement in the BBT group: median improvement of 1.2 units (range, 5.2 to – 1.6) in the BBT group compared with 0.4 units (range, 3.4 to – 2.9) in the control group (P = 0.09). These changes were spread fairly evenly across all four domains.
Discussion
In this largely self-selected cohort of asthma sufferers with high medication use, those randomised to BBT lowered their minute volume, reduced beta2-agonist use, and demonstrated a trend towards lower inhaled steroid doses and greater improvement in quality of life than did those randomised to a control breathing program. No change in lung function was noted.
To minimise the effect of environmental influences and to maintain blinding, BBT and control group classes ran simultaneously. This posed logistic problems not often encountered in asthma intervention studies, where subjects are usually enrolled sequentially over a substantial period of time, giving an opportunity to adjust treatment. The need to complete randomisation for all subjects before the interventions precluded comprehensive pre-study medication optimisation.
Conventional teaching argues that hyperventilation and hypocapnia are the result rather than the cause of airway narrowing in asthma. Thus, increases in ventilation are seen in normal subjects with methacholine-induced bronchoconstriction.11 On the other hand, hyperventilation may cause bronchoconstriction.12, 13 Gardner suggests that asthma and hyperventilation may interact through a process in which symptoms due to asthma and the consequent induced hyperventilation result in anxiety and further increases in minute volume.14
We could not show any change in ET CO2 in either BBT or control subjects; for both groups, ET CO2 remained significantly below that of normal individuals. MV was high (accepting a mean predicted resting MV of 5 L/min) in both BBT and control groups, and to a lesser (although statistically similar) extent in the normal subjects. Perhaps this may be explained in part by the use of a water-sealed spirometer, mouthpiece and noseclip, which have been shown to elevate tidal volume and respiratory rate.15 MV declined in the BBT group. Because MV and ET CO2 were measured at different times and on different devices, no direct correlation of MV and ET CO2 is possible.
What alternative explanations are there for the observed improvements with BBT? Both groups were contacted fortnightly by the research team. In addition, some of the BBT subjects who were experiencing difficulties with the technique were contacted frequently by the Buteyko therapist. We did not anticipate this contact, which leaves the study open to the criticism that the BBT group were influenced in ways the control group were not. The study clearly would have been stronger with matched, controlled phone contact between both BBT and control practitioners and subjects.
BBT might also have altered subjects’ perceptions of asthma severity without affecting the underlying disease. This could account for the reduction in medication use and trends toward improvements in quality of life, and is consistent with the absence of any change in objective measures of airway calibre. On the other hand, the reduction in medication use in the BBT group did not lead to a decline in lung function, and rates of oral steroid use and hospital admission were similar in each group.
Reduction in beta2-agonist use itself might have led to an improvement in asthma control. There are suggestions that excess beta2-agonist use may adversely affect asthma control.16 To our knowledge, no study has suggested the converse, that reduction in beta2-agonist use may improve asthma control.
In conclusion, we found that those practising BBT reduced hyperventilation and their use of beta2-agonists. A trend toward reduced inhaled steroid use and better quality of life was observed in these patients without changes in objective measures of airway calibre.
Acknowledgements
A grant from the Australian Association of Asthma Foundations and assistance from Buteyko Australia supported the study. Ms Tess Graham of Buteyko Australia conducted the Buteyko breathing classes and reviewed the manuscript. Ms Jeanette Martin undertook the control group classes. The technical assistance of Mr Andrew Coates; the administrative help of Mr John Laing; and the assistance of the staff of the Asthma Foundation of Queensland are gratefully acknowledged.
- Stalmatski A. Freedom from asthma: Buteyko’s revolutionary treatment. Hale Clinic Health Library. London: KyleCathie Ltd, 1997; 175.
- Singh V, Wisniewski A, Britton J, Tattersfield A. Effects of yoga breathing exercises (pranayama) on airway reactivity in subjects with asthma. Lancet 1990; 335: 1381-1383.
- Jain SC, Talukdar B. Evaluation of yoga therapy programme for patients of bronchial asthma. Singapore Med J 1993; 34: 306-308.
- Girodo M, Ekstrand KA, Metivier GJ. Deep diaphragmatic breathing: rehabilitation exercises for the asthmatic patient. Arch Phys Med Rehabil 1992; 73: 717-720.
- Renfroe KL. Effects of progressive relaxation on dyspnea and state anxiety in patients with chronic obstructive pulmonary disease. Heart Lung 1988; 17: 408-413.
- Gibson PG, Wlodarczyk JH, Borgas T. Drug delivery in asthma: a comparison of spacers with a jet nebuliser. Aust N Z J Med 1995; 25: 324-329.
- Smyth ET, Pavord ID, Wong CS, et al. Interaction and dose equivalence of salbutamol and salmeterol in patients with asthma. BMJ 1993; 306: 543-545.
- Marks GB, Dunn SM, Woolcock AJ. A scale for the measurement of quality of life in adults with asthma. J Clin Epidemiol 1992; 45: 461-472.
- Marks GB, Dunn SM, Woolcock AJ. An evaluation of an asthma quality of life questionnaire as a measure of change in adults with asthma. J Clin Epidemiol 1993; 46: 1103-1111.
- Statview [computer program]. Version 4.1. Cary, North Carolina: SAS Institute, 1995.
- Chadha TS, Schneider AW, Birch S, et al. Breathing pattern during induced bronchoconstriction. J Appl Physiol 1984; 56: 1053-1059.
- Gayrard P, Orehek J, Grimaud C, Charpin J. Bronchoconstrictor effects of a deep inspiration in patients with asthma. Am Rev Respir Dis 1975; 111: 433-439.
- Van den Elshout FJ, van Herwaarden CL, Folgering HTM. Effects of hypercapnia and hypocapnia on respiratory resistance in normal and asthmatic subjects. Thorax 1991; 46: 28-32.
- Gardner WN. The pathophysiology of hyperventilation disorders. Chest 1996; 109: 516-534.
- Gilbert R, Auchincloss JH Jr, Brodsky J, Boden W. Changes in tidal volume, frequency, and ventilation induced by their measurement. J Appl Physiol 1972; 33: 252-254.
- Taylor DR, Sears MR, Herbison GP, et al. Regular inhaled beta agonists in asthma: effects on exacerbations and lung function. Thorax 1993; 48: 134-138.
(Received 11 Aug 1997, accepted 18 Aug 1998)
Authors’ details
Mater Adult Hospital, South Brisbane, QLD.
Simon D Bowler, FRACP, Thoracic Physician.
Brompton Hospital, South Kensington, London, UK.
Amanda Green, GradDipPhys, Physiotherapist.
Department of Medicine, University of Queensland, Princess Alexandra Hospital, Woolloongabba, QLD.
Charles A Mitchell, FRACP, Associate Professor.
Reprints: Dr S D Bowler, Director, Respiratory Medicine, Mater Adult Hospital, South Brisbane, QLD 4101.
Email: sbowler@mater.org.au
